- Home
- Oral Hormones
- Aciclovir
- Acid Ascorbic
- Alendronat
- Ambroxol
- Anadrol 50
- Anapolon
- Anastrozol
- Anavar 10
- Chlorambucil
- Cialis
- Citomed
- Clemastin 10
- Clenbuterol
- Clomed
- Clomid
- Danabol 10
- Desloratadin
- Dianabol 10
- Domperidon 10
- Doperidon 10
- Enalapril
- Esculap
- Exedrol
- Femara
- Finasterida 5
- Halotest
- Izoniazida
- Leflunomide
- Letrozol
- Losartan
- Melfalan
- Micofenolat 250
- Nolvadex
- Ofloxacin 200
- Oxandrolon
- Pemidal
- Primobol
- Procarbazine
- Provimed
- Proviron
- Risperidon
- Salbutamol
- Spironolaction 25
- Stromba 50
- Tamoximed
- Tiomax 600
- Trienol
- Turanabol
- Turanabol 10
- Viagra 100
- Winstrol 10
- Zaleplon
- Injectable Hormones
- Boldenone Undecylenate
- Masteron
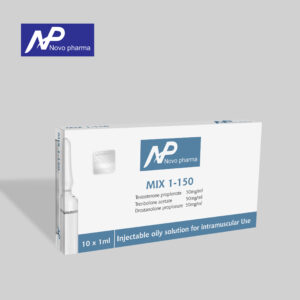
- MIX 1-150
- MIX 2-225
- Nandrolone Decanoate
- Nandrolone Phenylpropionate
- Parabolan
- Primobolan
- Stanozolol
- Sustanon
- Testosterone Cypionate
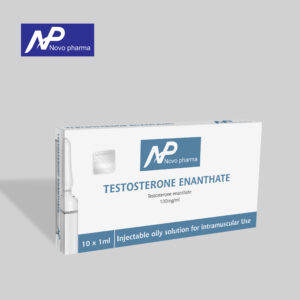
- Testosterone Enanthate
- Testosterone Phenylpropionate
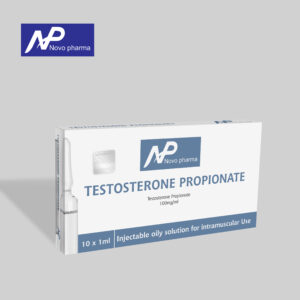
- Testosterone Propionate
- Testosterone Suspension
- Tren mix
- Trenbolone Acetate
- Trenbolone Enanthate
- Peptides
- Sarms
- Product Verification
- Contact Us